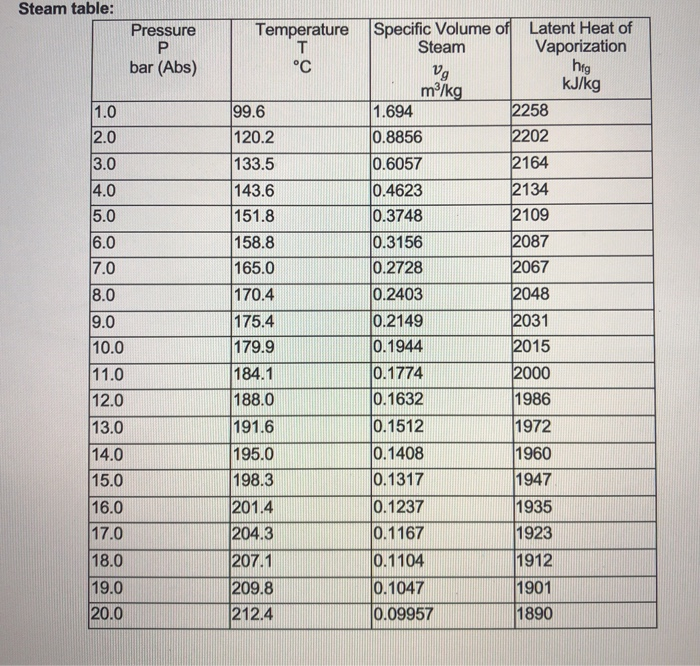
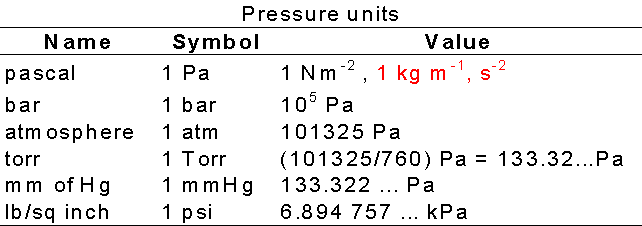
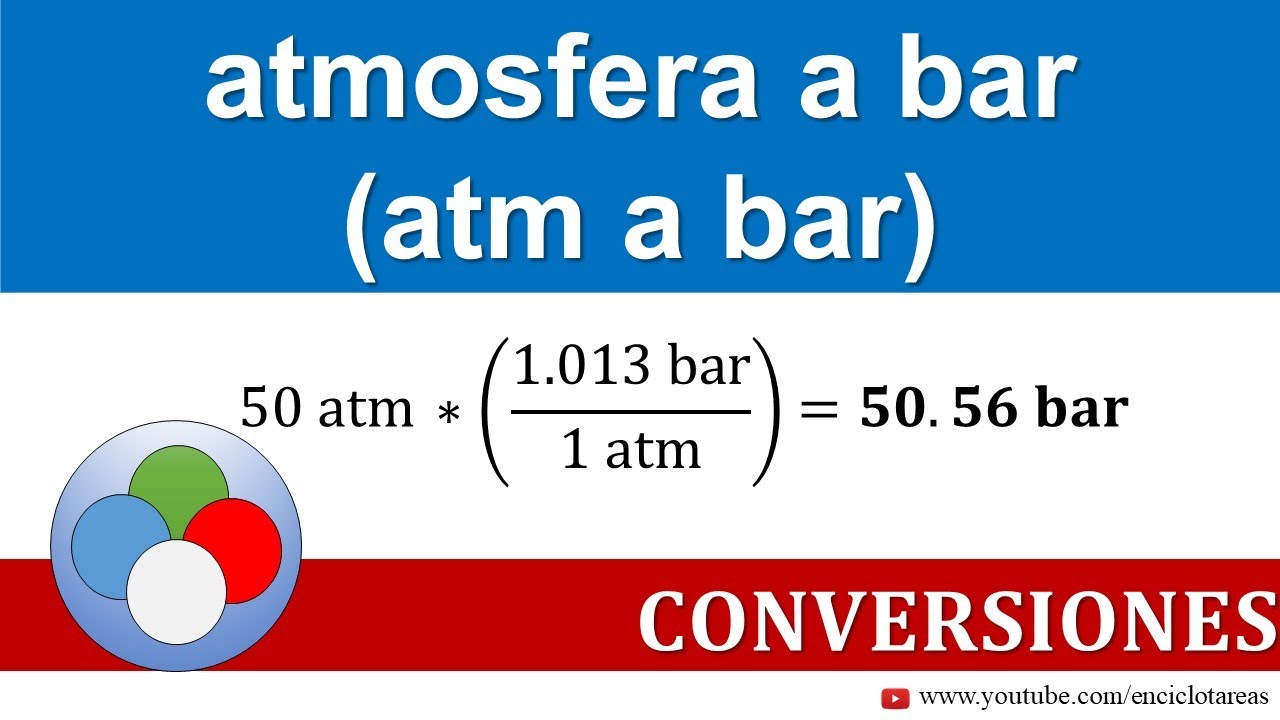
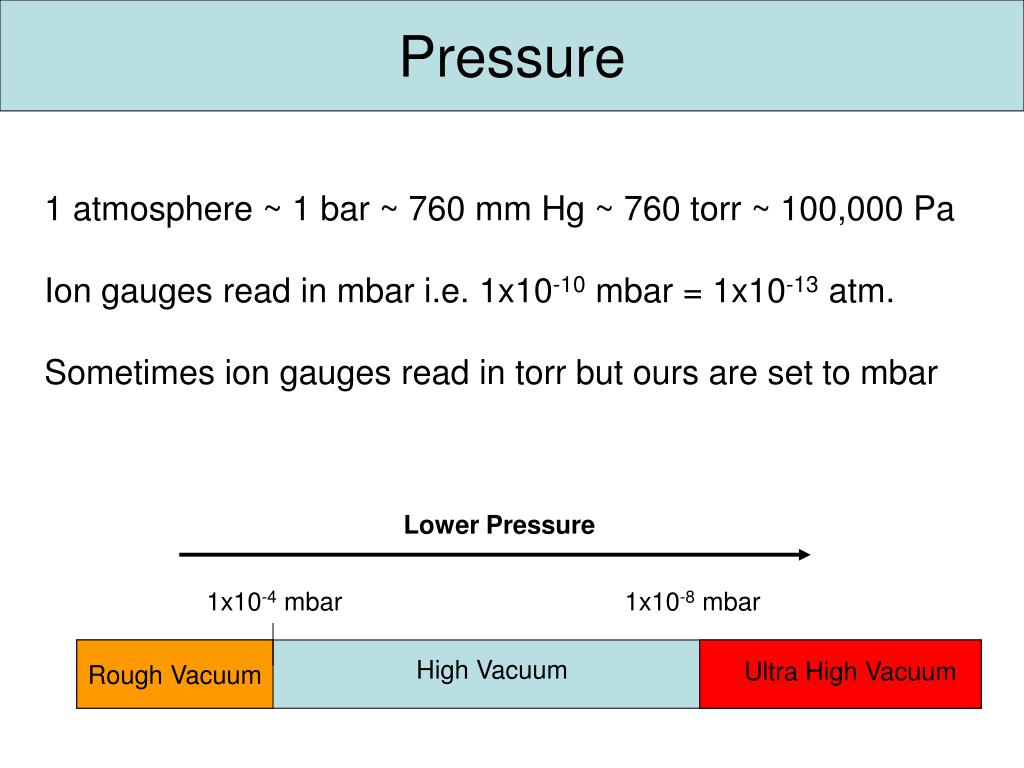
pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

SOLVED: [Place the following units of pressure In order from lowest to highest pressure. bar mm Hg < 1 Pa < 1 atm atm Pa K mm Hg < 1 bar ci

pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

SOLVED: If 1 bar = 1.01325 atm, and the atmospheric pressure at the top of Mount Everest is 0.332 atm, what is the pressure in bar? Express your answer to three significant figures.
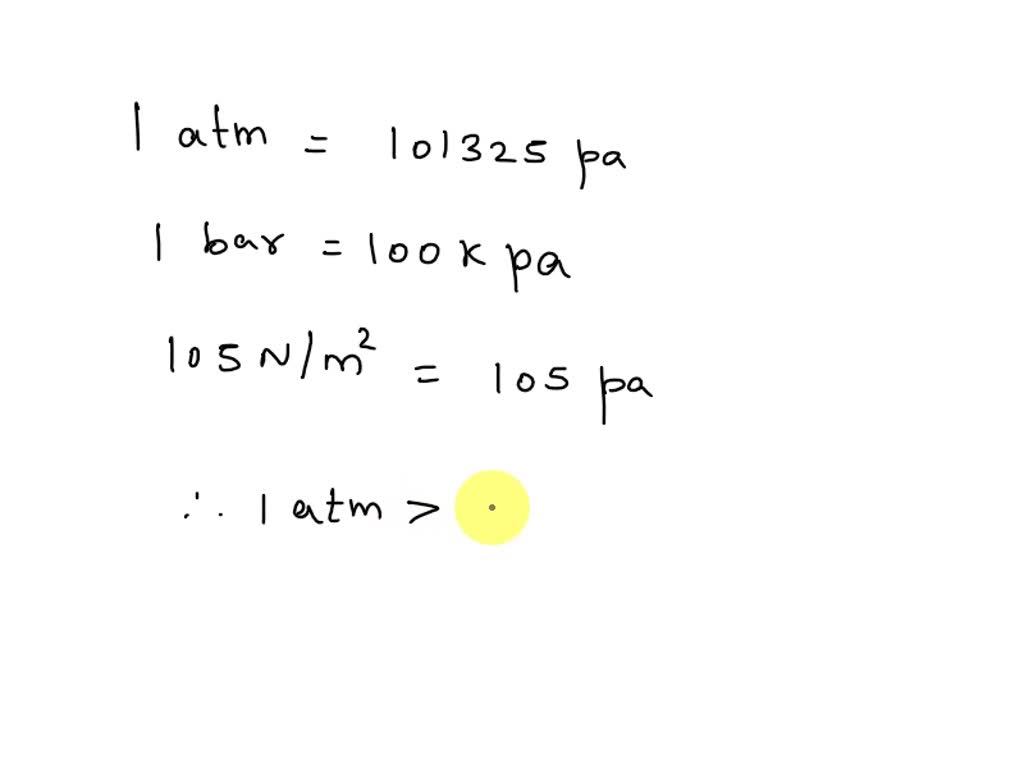
SOLVED: Which is of the highest value? (a) 1 bar (b) 105 N/m2 (c) 1 atm (d) 100 kPa (e) None of these


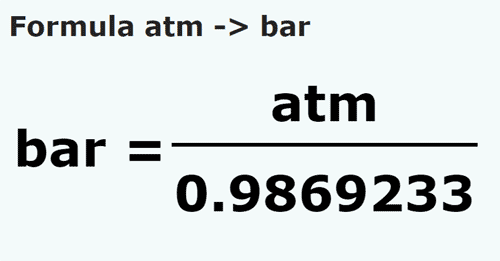
:max_bytes(150000):strip_icc()/GettyImages-126332621-56a133a93df78cf7726859c3.jpg)
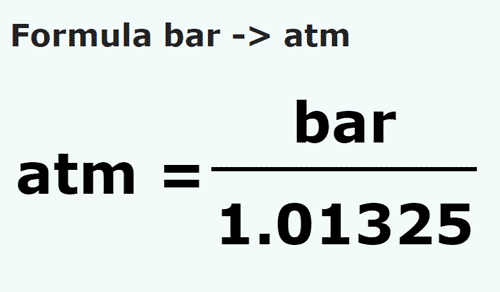

:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)