Which of the following molecules does not have a dipole moment? a. HI b. H2O c. C2H2 d. CH3CH2OH | Homework.Study.com

Question Video: Determining Whether Some Common Simple Molecular Compounds Should Be Classified As Polar or Nonpolar Solvents | Nagwa

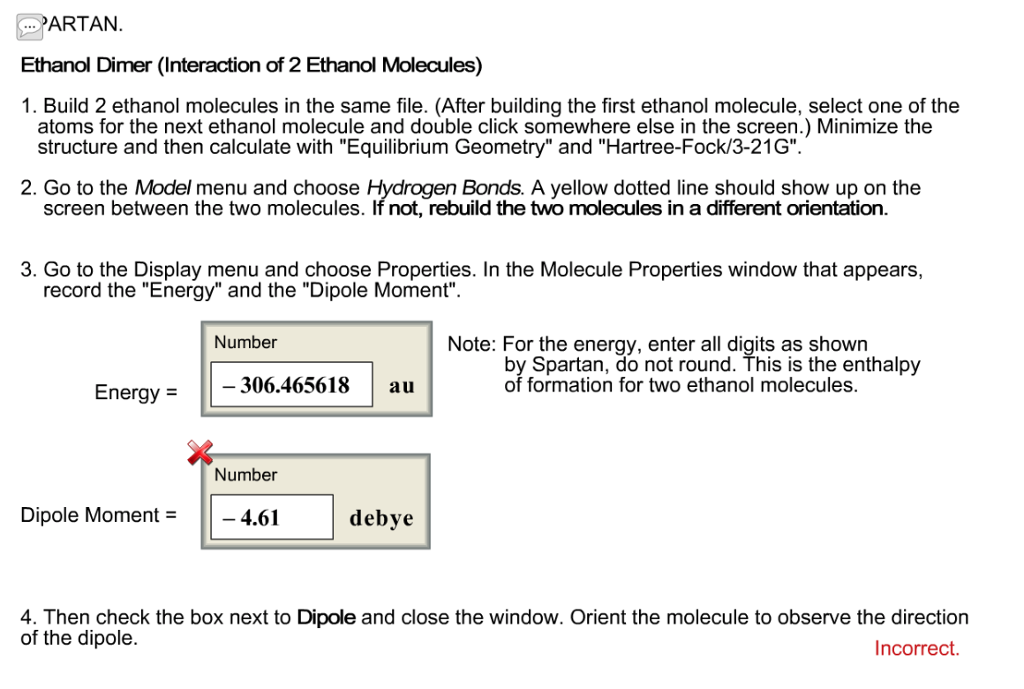
Predict the direction of the net molecular dipole for ethanol, as well as predict the shape around each carbon and nitrogen atom in dimethylformamide, and predict whether the molecule is polar or

Predict the direction of the net molecular dipole for ethanol, as well as predict the shape around each carbon and nitrogen atom in dimethylformamide, and predict whether the molecule is polar or
Alcohols Alcohol – any organic compound containing a hydroxyl (R-OH) group Uses: synthetic intermediate, cleanser, cosm

Molecular Dynamics Investigation of the Influence of the Hydrogen Bond Networks in Ethanol/Water Mixtures on Dielectric Spectra | The Journal of Physical Chemistry B

3) NUZ 120. The dipole moment is the highest (1) Trans-2-butene . (2) 1,3-dimethylbenzene B) Acetophenone (4) Ethanol
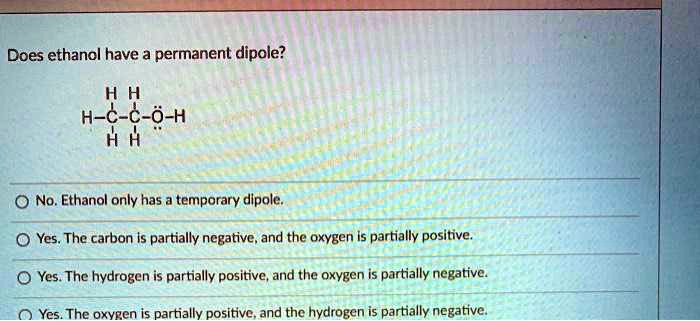
SOLVED: Does ethanol have a permanent dipole? H-C-C-O-H H H No. Ethanol only has a temporary dipole. Yes. The carbon is partially negative, and the oxygen is partially positive. Yes. The hydrogen
Chemical structure, dipole moment δ, in D, and Gutmann's donor number... | Download Scientific Diagram