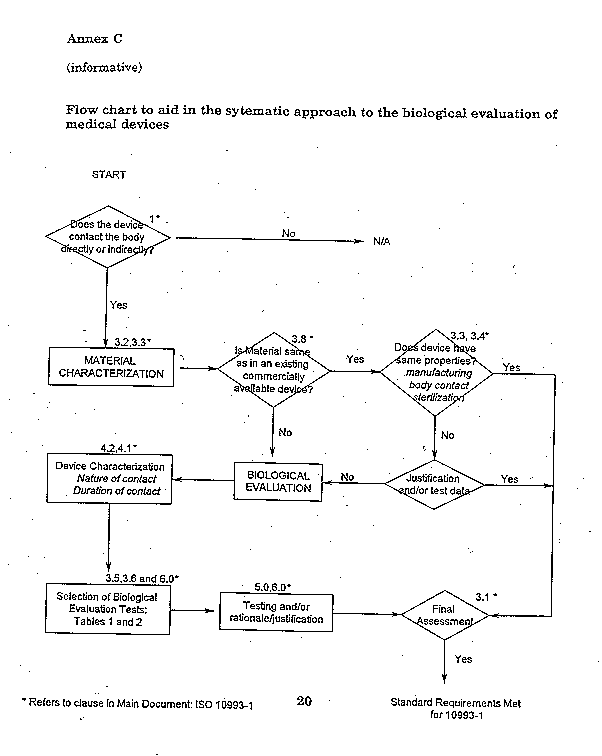
ISO 10993-1:2018 - Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process

Biological assessment of medical devices according to the ISO 10993-1 standard - GMED Medical Device Certification

ANSI/AAMI/ISO 10993-1:2009/(R)2013 Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process (Paperback): AAMI: Amazon.com: Books

Table I from Towards a logic-based extension of a relational software tool for coherent technical documentation of medical devices | Semantic Scholar

nueva edición norma ISO 10993-1:2018 biocompatibilidad con cambios relevantes | Red de Tecnologías Sanitarias y Productos Sanitarios

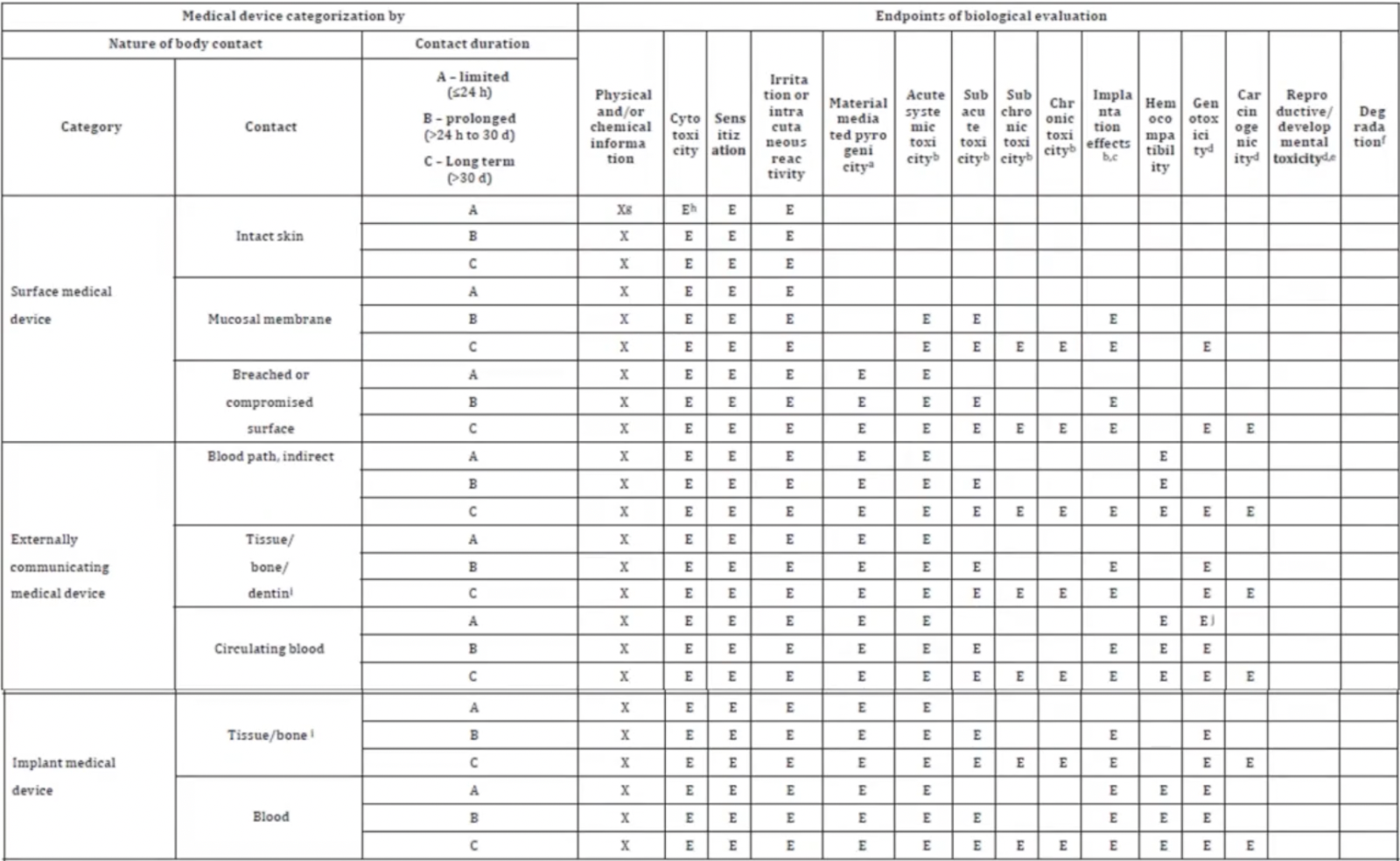
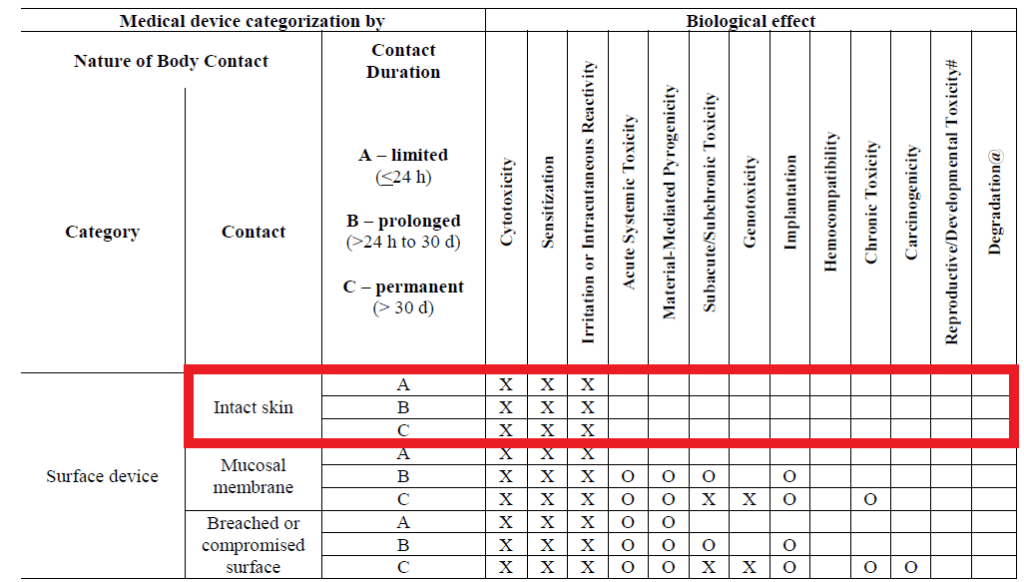
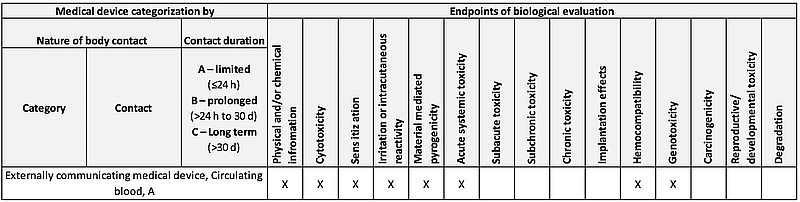

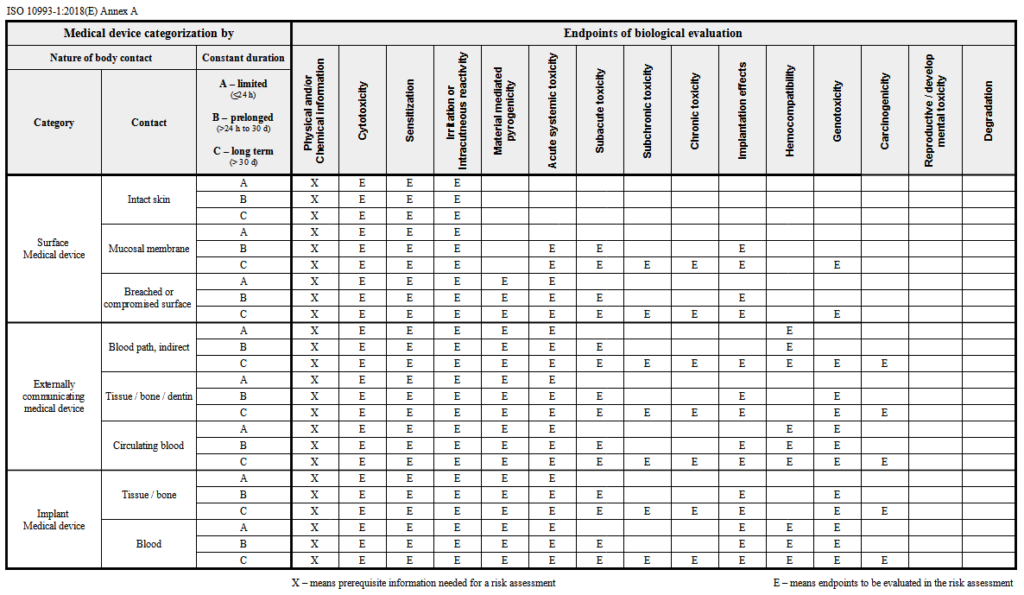


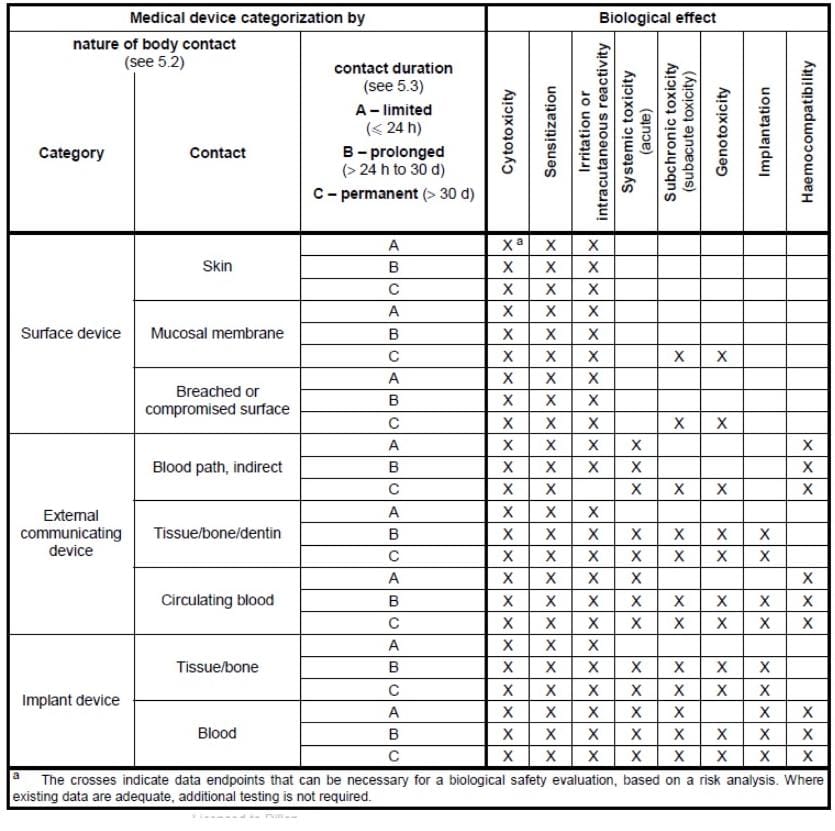

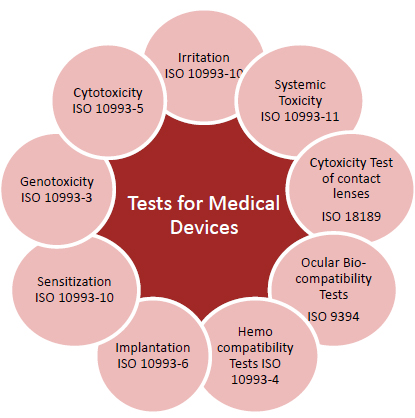
![Medical device - Biological Evaluation [ISO 10993] - WBR LABS Medical device - Biological Evaluation [ISO 10993] - WBR LABS](https://wbrlabs.in/wp-content/uploads/2020/10/Biological-Evaluation-of-Medical-device.jpg)