Efficacy of covid-19 vaccines in immunocompromised patients: systematic review and meta-analysis | The BMJ

Covid autumn booster vaccine 2023: Everything you need to know - Department of Health and Social Care Media Centre
Factsheet: COVID-19 Vaccine Guidance for People Living with Multiple Sclerosis (English, Spanish) | Vaccine Resource Hub

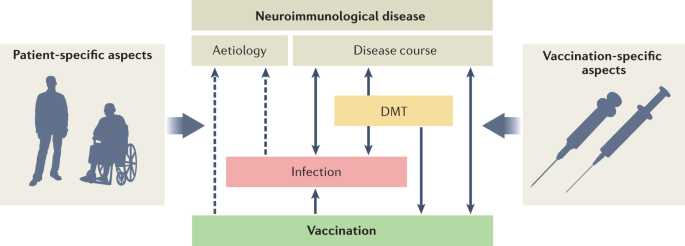
Expert Perspectives on COVID-19 Vaccination for People Living with Multiple Sclerosis | Neurology and Therapy

Breakthrough COVID-19 in Patients With Multiple Sclerosis: Snapshot From One Data Registry – Consult QD
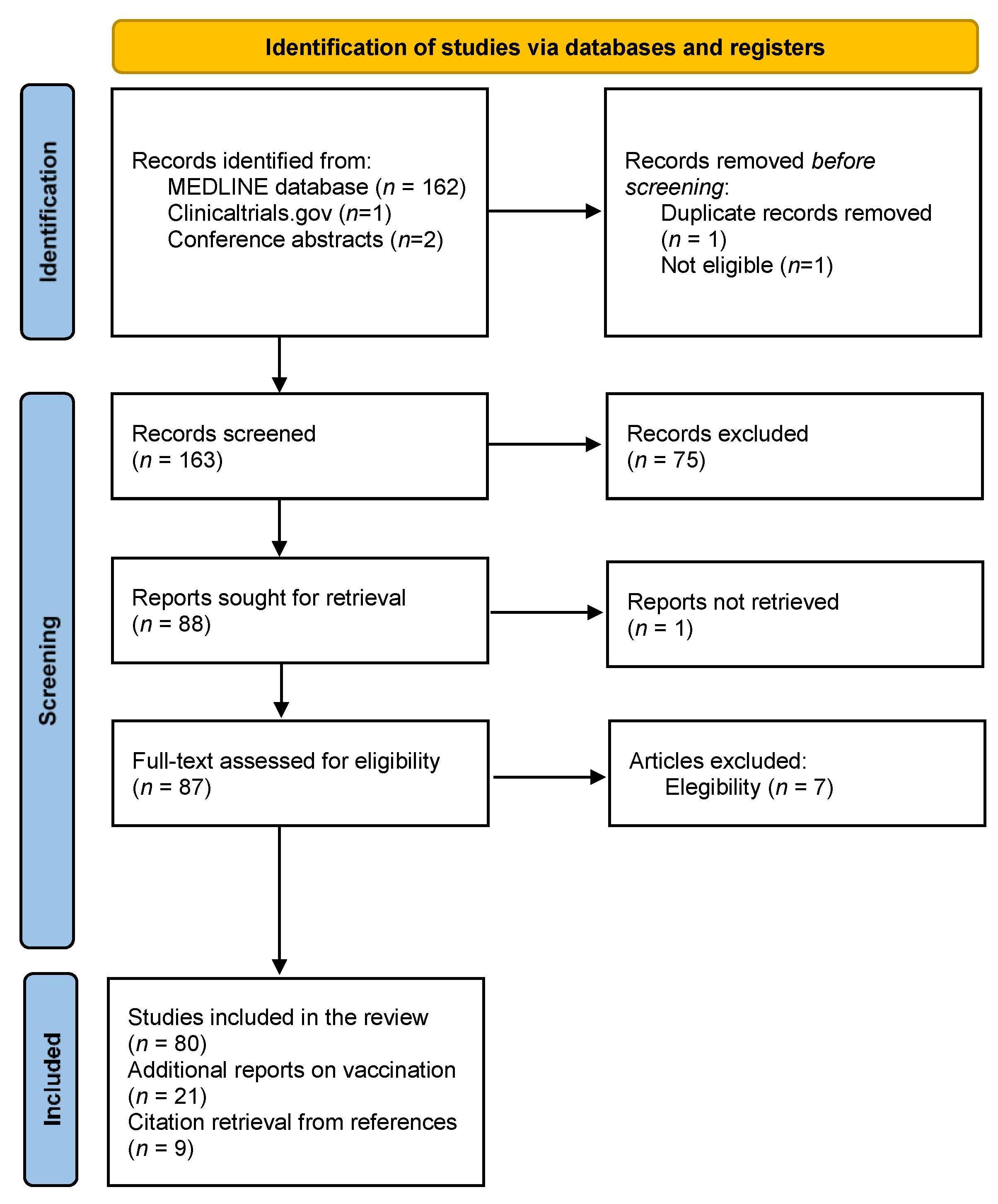
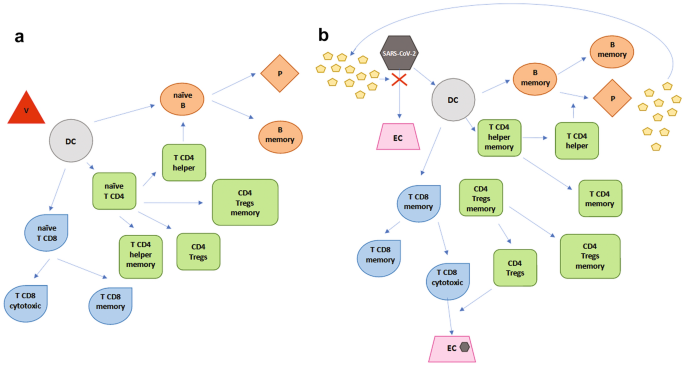
Vaccines | Free Full-Text | Multiple Sclerosis, Disease-Modifying Therapies and COVID-19: A Systematic Review on Immune Response and Vaccination Recommendations
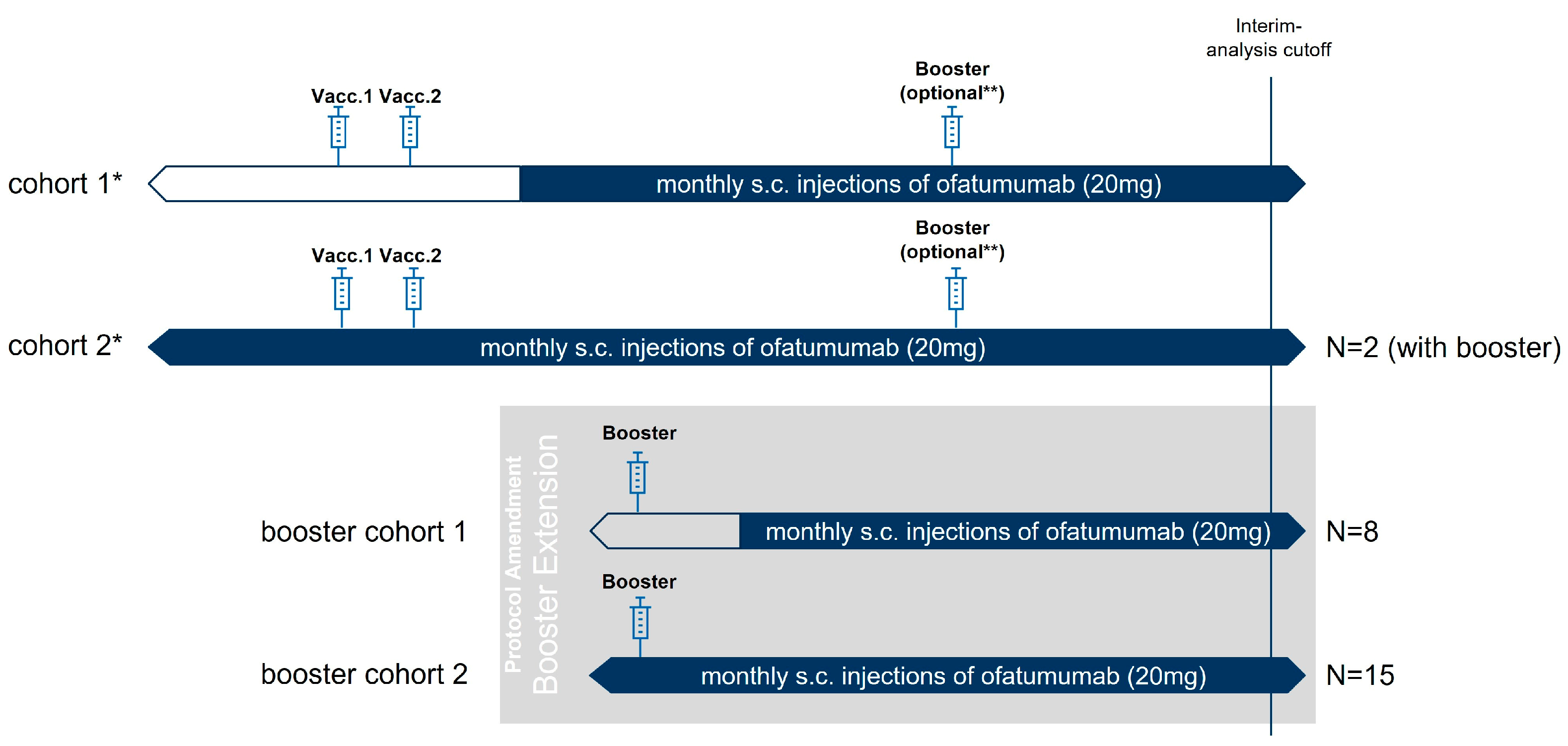
Vaccines | Free Full-Text | Results on SARS-CoV-2 mRNA Vaccine Booster from an Open-Label Multicenter Study in Ofatumumab-Treated Participants with Relapsing Multiple Sclerosis

Multiple sclerosis disease-modifying therapies and COVID-19 vaccines: a practical review and meta-analysis | Journal of Neurology, Neurosurgery & Psychiatry

COVID-19 | Humoral response and safety of the third booster dose of BNT162b2 mRNA COVID-19 vaccine in patients with multiple sclerosis treated with ocrelizumab or fingolimod | springermedizin.de

Humoral immune response in multiple sclerosis patients following PfizerBNT162b2 COVID19 vaccination: Up to 6 months cross-sectional study - ScienceDirect